
Apple has started sending out notifications to users of its Apple Watch announcing the commencement of its "Heart Survey", the media reported.
In November 2017, Apple had asked the users of its Apple Watch if they wanted to take part in a study called the "Apple Heart Survey" being conducted by the technology giant in collaboration with Stanford Health.
The project is designed to further ongoing efforts in the health sciences market, AppleInsider reported late on Saturday.
Tapping on the notification opens the Heart Study app after which the users will be directed to verify whether they are "comfortable" speaking and writing in English, and have not been diagnosed with atrial fibrillation or atrial flutter -- irregular heart rhythms.
Once a user confirms that they meet these specified requirements, Apple sends out a pair of emails containing an informed consent document and information regarding HIPAA rights and regulations.
The data from the survey will be used to develop new product technologies, the report said.
"This study is part of the development of a new investigational device and certain Study Data will be used for US Food and Drug Administration (FDA) submission to seek approval of the investigational device," Apple said in a statement.
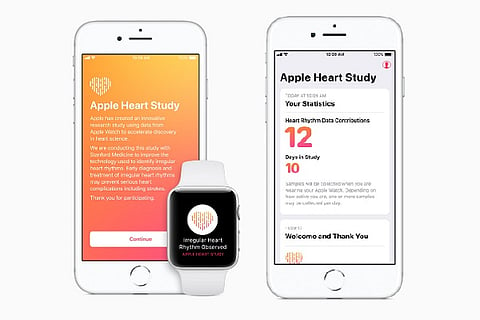
Apple Watch has a specialised heart rate sensor and a dedicated app to collect data that can be used to identify irregular heart rhythms.
If a user displays abnormal heart activity, they receive a notification on their Apple Watch and iPhone, a free consultation with a study doctor and an electrocardiogram peripheral for additional monitoring.
How can people join the study?
The study is currently open to US residents above the age of 22, using an iPhone 5s or later with iOS 11 and an Apple Watch Series 1 or later with watchOS 4.
The users need to download the app, give consent for participation and the heart rate sensor data they provide over time will be collected by Stanford Medicine and Apple for research analysis.
The process:
The Apple Heart Study app passively monitors your heart rate along with your heart rhythm. If an irregular heart rhythm — also known as an arrhythmia — is identified, the app will notify the user after which they will receive a free video consultation on their iPhone with the study’s medical professionals for further analysis.
In some cases, the doctor may recommend a BioTelemetry electrocardiogram (ECG) patch for additional monitoring. The patch will be mailed at no cost and should be worn for seven days. The ECG data gathered over this period will be used to determine whether atrial fibrillation (AFib) or another type of irregular heart rhythm is present.
With IANS inputs